A foundation work of modern molecular physics
Over de continuiteit van de gas- en vloeistoftoestand. Academisch Proefschrift . . . aan de Hoogeschool te Leiden.
Leiden: A. W. Sijthoff, 1873.
1st Edition. Hardcover. Near Fine. Item #003104
8vo (220 x 155 mm). viii, 134, [2] pp., including half-title, one folding lithographed plate, and errata leaf. 20th-century half calf over marbled boards, spine lettered and decorated in gilt (light rubbing to extremities). Original printed wrappers bound in. Faint spotting on the plate, wrappers slightly dust-soiled, but otherwise a fresh and bright copy. Provenance: Torsten Malmberg (bookplate to front pastedown); illegible inscription on half-title. ----
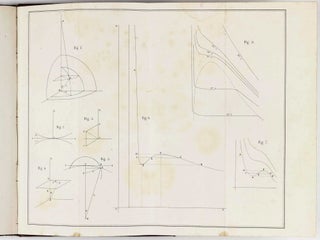
DSB XIV, p.109-11; Stanitz 80; not in Norman. EXCEPTIONALLY RARE FIRST EDITION of Van der Waals' doctoral dissertation on gas and fluid states which laid the foundations for modern molecular physics, a work which, in the words of James Clerk Maxwell, "at once put his name among the foremost in science." Van der Waals, the son of a carpenter, was because of his modest circumstances a relatively slow developer as a professional scientist: he returned to university at the age of thirty, after years spent as a primary and secondary-school teacher. Shortly after the appearance of his dissertation, however, he was promoted to the chair in physics at the newly founded university of Amsterdam, where he was succeeded, on his retirement in 1907, by his son. In 1910 he was awarded the Nobel Prize in Physics. "On the basis of his knowledge of the work of Clausius and other molecular theorists, he wrote his dissertation, 'Over de continuiteit' ... Using rather simple mathematics, the dissertation gave a satisfactory molecular explanation for the phenomena observed in vapors and liquids by Thomas Andrews and other experimenters... This was one of the first descriptions of a collective molecular effect" (DSB). At the time van der Waals wrote his thesis, the molecular structure of fluids had not been accepted by most physicists, and liquid and vapor were often considered as chemically distinct. It was this thesis that affirmed the reality of molecules and allowed an assessment of their size and attractive strength. His new formula revolutionized the study of equations of state. By comparing his equation of state with experimental data, Van der Waals was able to obtain estimates for actual molecule sizes and the strength of their mutual attraction. The effect of Van der Waals' work on molecular physics in the 20th century was direct and fundamental. By introducing parameters characterizing molecular size and attraction in constructing his equation of state, Van der Waals set the tone for modern molecular science. This work is a great rarity with no copy recorded at auction in the past 20 years. - Visit our website for additional images and information.
Price: 8,500 € * convert currency
Delivery time up to 10 days. For calculation of the latest delivery date, follow the link: Delivery times
Lieferzeit max. 10 Tage. Zur Berechnung des spätesten Liefertermins siehe hier: Lieferzeiten